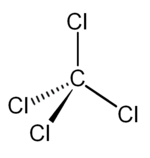
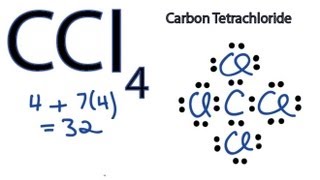
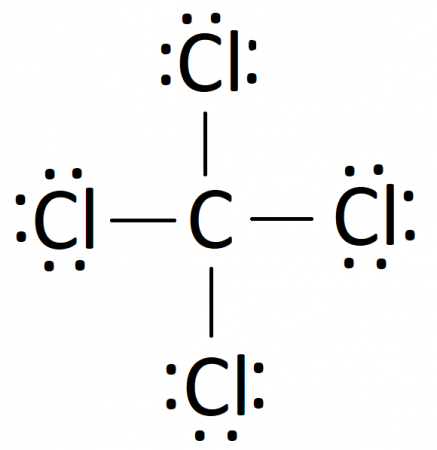Carbon Tetrachloride (CCl4); Lewis Structure, Molecular Geometry, Polarity, And Applications | Scientific Sarkar

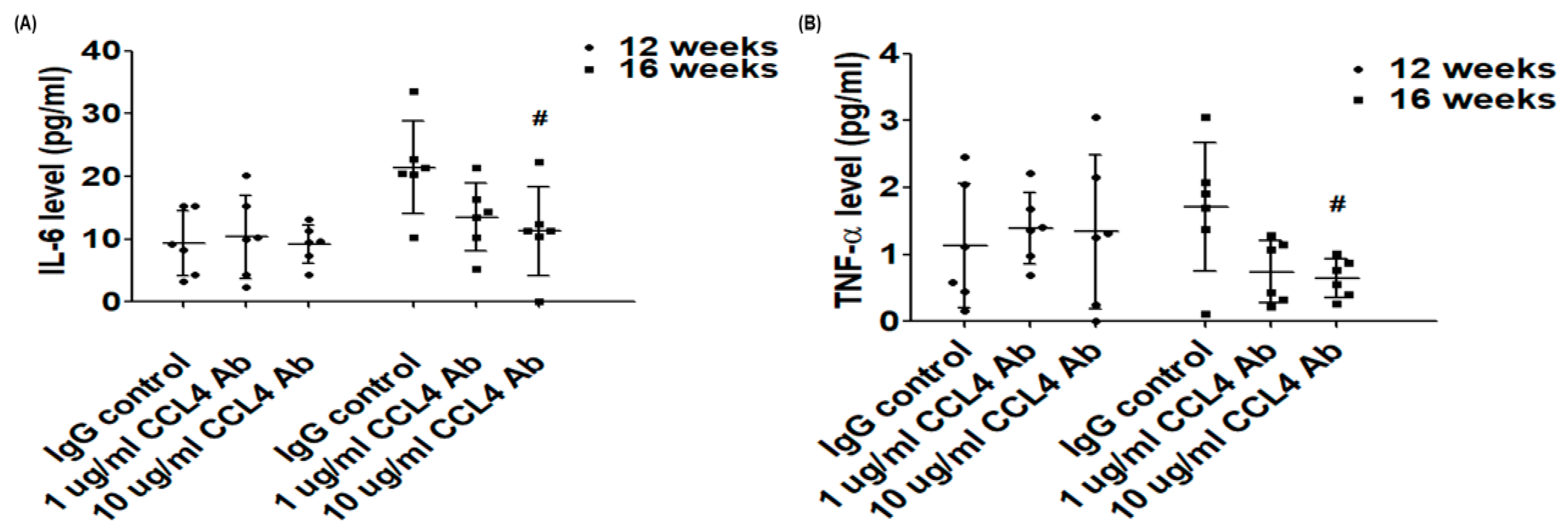
Biomedicines | Free Full-Text | CCL4 Deletion Accelerates Wound Healing by Improving Endothelial Cell Functions in Diabetes Mellitus

Draw the Lewis dot structure for CCl4. Determine the electron geometry and molecular shape of this molecule. Is this molecule polar or nonpolar? | Homework.Study.com

Provide the following information for CCl4. a. Lewis dot structure b. bond polarity (show dipole vectors) c. molecular polarity d. identify all intermolecular forces present | Homework.Study.com

El Tetracloruro De Carbono Es El Compuesto Inorgánico Con La Fórmula Ccl4. Antes Fue Ampliamente Utilizado En Los Extintores De Incendios, Como Un Precursor De Los Refrigerantes, Y Como Agente De Limpieza.

The necessary steps required to show the formation of CCl4 by Lewis electron dot diagram has been jumbled. Arrange them in a sequence.(a) Thus, an electron pair is shared between C and

Draw the Lewis dot structure for CCl4. Determine the electron geometry and molecular shape of this molecule. Is this molecule polar or nonpolar? | Homework.Study.com

El tetracloruro de carbono es el compuesto inorgánico con la fórmula CCl4. Anteriormente se utilizaba ampliamente en extintores de incendios, como precursor del refrigerante Fotografía de stock - Alamy